加热碱式碳酸铜方程式

碱式碳酸铜加热分解方程
图片尺寸1280x720
碱式碳酸铜加热 2.氢氧化铁中加入稀硫酸
图片尺寸486x648
碱式碳酸铜cu2oh2co3是孔雀石的主要成分俗称铜绿受热可分解生成氧化
图片尺寸520x464
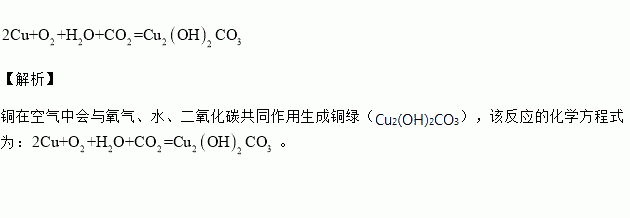
熟记 个化学方程式: 2cu o2 h2o co2=== cu2(oh)2co3(铜锈), 3cu 8
图片尺寸1080x810
a.当发现家中的煤气灶漏气时应关闭进气阀门,开窗通风
图片尺寸447x188
加热碱式碳酸铜:cu2(oh)2co3 加热 2cuo h2o co2↑ 15.加热氯
图片尺寸537x767![该"绿锈"俗称"铜绿",又称"孔雀石"[化学式为cu2(oh)2co3],铜绿](https://imgs.wantubizhi.com/img/ADDA3C5D760C6828B77B505A29D43E833C8594615DAAD04D7F07C57A975C5016B6B41351DB4C66D0D04A08E3D14F05458DDF9206EAEA8E53E2828EE1459D83B850FD2894F568A3D6273CAABE90EBF9AAE562A17D9554C368F34F85F385B67720)
该"绿锈"俗称"铜绿",又称"孔雀石"[化学式为cu2(oh)2co3],铜绿
图片尺寸299x281
碱式碳酸铜受热分解除了生成氧化铜,还生成了________.
图片尺寸418x146
反应的化学方程式为 , 充分加热30g含氧化铜的碱式碳酸铜
图片尺寸630x301
展开全部 和空气中的氧气,二氧化碳反应生成碱式碳酸铜 1 已赞过 已
图片尺寸372x87
(1)碱式碳酸铜的相对分子质量是 _百度
图片尺寸627x294
第36个化学方程式,谢谢!
图片尺寸816x468
加热碱式碳酸铜:cu2(oh)2co3 加热 2cuo h2o co2↑ 15.加热氯
图片尺寸538x767
1,碱式碳酸铜加热分解生产氧化铜,水和二氧化碳; 2,水通电分解成氢气
图片尺寸1600x1200
写出下列反应的表达式
图片尺寸630x387
反应的化学方程式为 ,充分加热24g含氧化铜的碱式碳酸铜固体.
图片尺寸630x210
请写出该反应的化学方程式: .
图片尺寸630x218![(3)加热铜绿 .(4)用赤铁矿炼铁 .(5)面粉的化学式为[c6h10o5]n](https://imgs.wantubizhi.com/img/735A5D1F980B52A636CC46F12F2D1A739920464DF1ACD5D590597B22F51F0277E6CC4998C50E03FB2B58DCA7EAE3188E3F1CCFFA6D83825FAFEF8CAEC786ED41C919C829CAB09EE52F3AED5B10223EE8FE9695CE9350C60F286FF733F48AA9C3)
(3)加热铜绿 .(4)用赤铁矿炼铁 .(5)面粉的化学式为[c6h10o5]n
图片尺寸420x388
受热可生成黑色氧化铜固体,二氧化碳和水,如图是加热碱式碳酸铜的实验
图片尺寸206x184
九年级上册化学第三单元思维导图氧化铁2绿色固体碱式碳酸铜3
图片尺寸596x479






![该"绿锈"俗称"铜绿",又称"孔雀石"[化学式为cu2(oh)2co3],铜绿](https://imgs.wantubizhi.com/img/ADDA3C5D760C6828B77B505A29D43E833C8594615DAAD04D7F07C57A975C5016B6B41351DB4C66D0D04A08E3D14F05458DDF9206EAEA8E53E2828EE1459D83B850FD2894F568A3D6273CAABE90EBF9AAE562A17D9554C368F34F85F385B67720)










![(3)加热铜绿 .(4)用赤铁矿炼铁 .(5)面粉的化学式为[c6h10o5]n](https://imgs.wantubizhi.com/img/735A5D1F980B52A636CC46F12F2D1A739920464DF1ACD5D590597B22F51F0277E6CC4998C50E03FB2B58DCA7EAE3188E3F1CCFFA6D83825FAFEF8CAEC786ED41C919C829CAB09EE52F3AED5B10223EE8FE9695CE9350C60F286FF733F48AA9C3)








![该"绿锈"俗称"铜绿",又称"孔雀石"[化学式为cu2(oh)2co3],铜绿](https://hiphotos.baidu.com/zhidao/pic/item/5882b2b7d0a20cf4d7b24cf372094b36acaf99f8.jpg)









![(3)加热铜绿 .(4)用赤铁矿炼铁 .(5)面粉的化学式为[c6h10o5]n](https://thumb.1010pic.com/pic6/api/STDAIMG_CZHX_2016072506052534654537_SYS201607250605337062495940_630.png)

