氮的价电子排布图
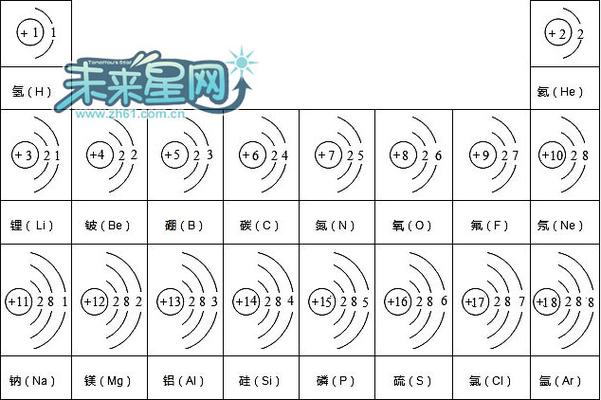
前18号元素(短周期元素)的核外电子排布图
图片尺寸634x422
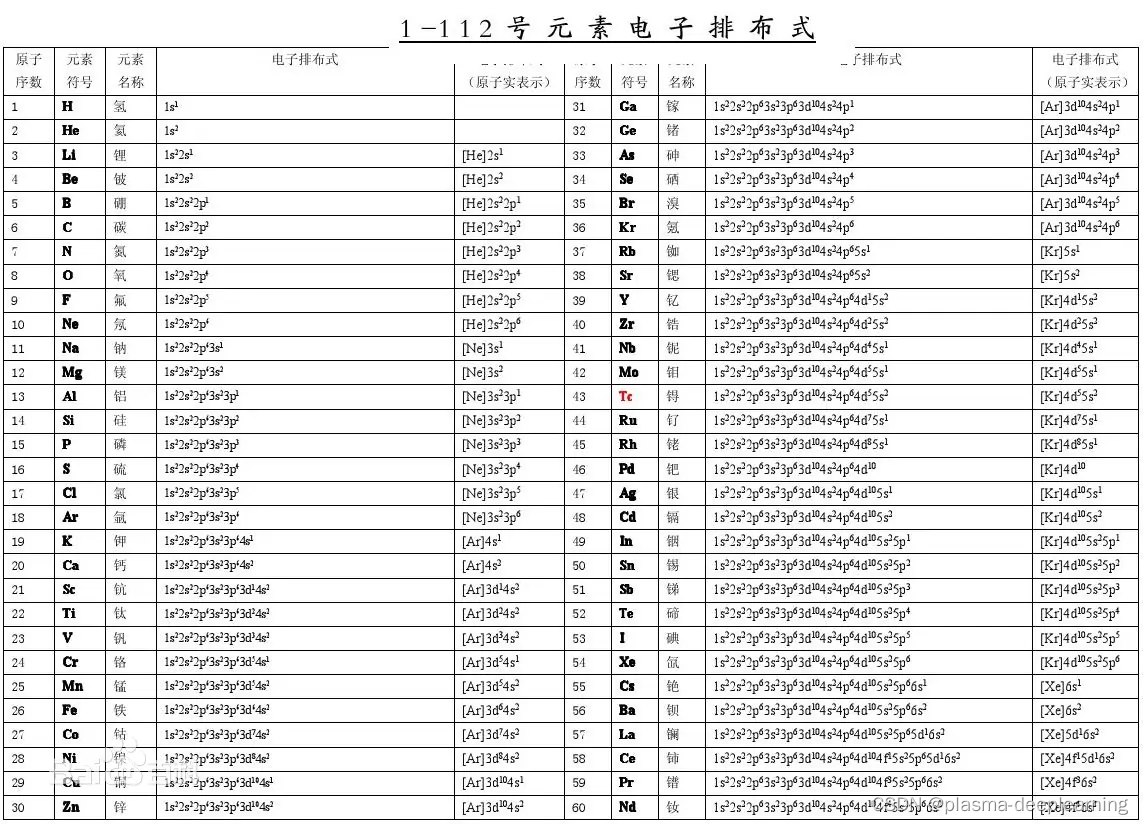
氮元素原子基态电子排布式
图片尺寸282x134
二,核外电子排布的方法
图片尺寸1143x824
氮及其化合物的"价类二维图"
图片尺寸337x223
s1个轨道,n 1p3个轨道 共九个轨道,9*2=18)都是为了使电子排布达到
图片尺寸1280x2276最外层电子数决定了什么
图片尺寸640x341氮元素价类二维图
图片尺寸946x717
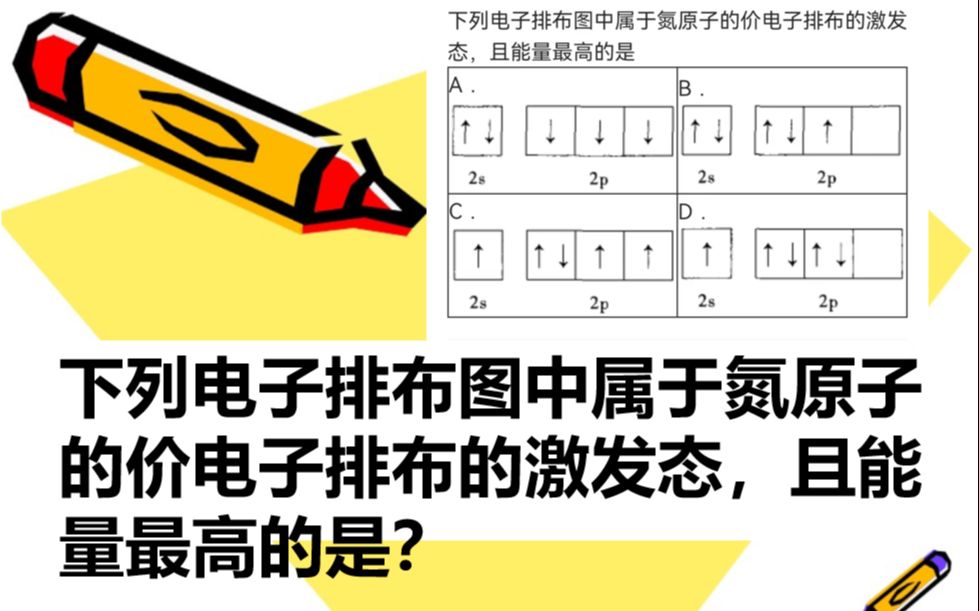
答案:a试题分析:基态原子电子排布必须遵循①能量最低原理,②泡利不
图片尺寸280x244
下列电子排布图中属于氮原子的价电子排布的激发态,且能量最高的是?
图片尺寸979x611
自然科学 物理 2能量最低原理,基态与激发态,光谱ppt 2,电子排布式
图片尺寸1080x810
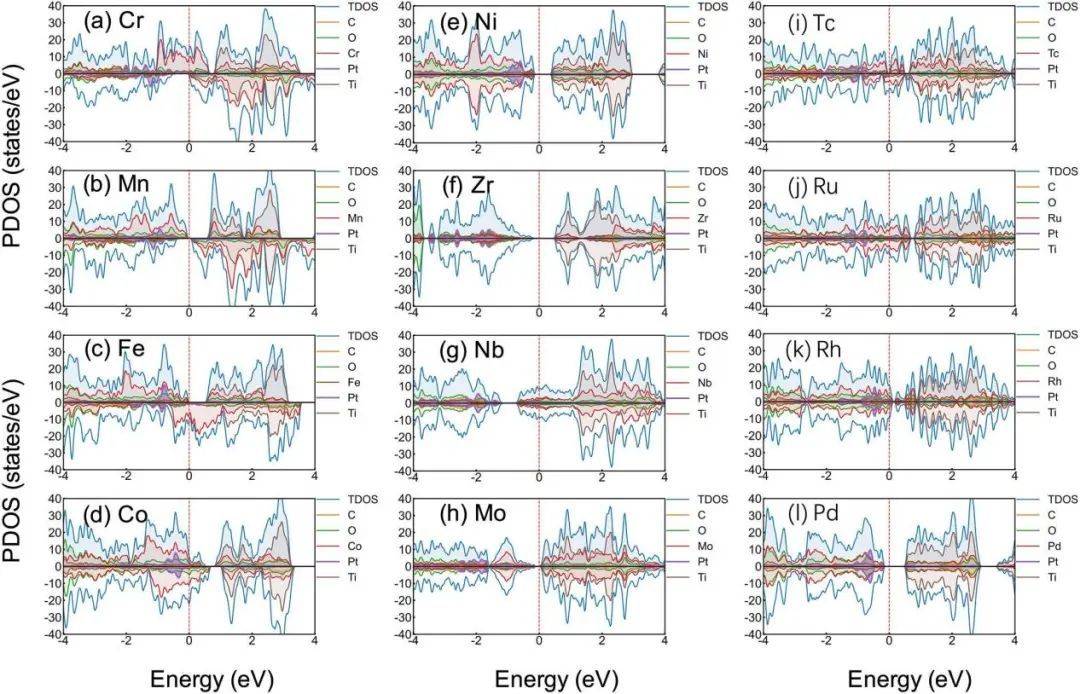
sci.:机器学习辅助探索影响orr/oer双功能催化活性_催化剂_原子_电子
图片尺寸1080x694
高考化学抢分秘籍-秘籍17物质结构与性质-练习1
图片尺寸586x605
常见氮原子的杂化类型
图片尺寸626x446
6. c是mg的基态原子的电子排布式,而a,b,d都不是基态原子的电子排布.
图片尺寸597x340
高考化学抢分秘籍-秘籍17物质结构与性质-练习1
图片尺寸586x968
(4)钠离子核外电子排布与 (填元素符号)原子
图片尺寸467x223
nacl的形成过程是:c.ch4的分子结构模型是:b.nh3的电子式是:a.
图片尺寸550x489微电子加速迈向后摩尔时代!
图片尺寸960x668多晶硅:晶硅光伏核心原材料
图片尺寸640x306
实力出圈!广工科研成果上新啦!
图片尺寸640x160
猜你喜欢:N的价电子排布图价电子排布图怎么画氮元素的电子排布图镍的原子结构示意图钒的价电子排布图钠的价电子排布图价电子排布式和排布图铜原子的价电子排布图铁的价电子排布图铜的价电子排布图锌的价电子排布图二价铁离子电子排布图cu的价电子排布图铬的价电子排布式图元素价电子排布图价电子排布图sc价电子排布图铁的电子排布图价层电子排布图cu的价层电子排布图原子电子层排布规律图价电子排布式氮原子的电子式图片价电子排布图画法铁的电子排布式氮原子的电子式电子排布图cu的价层电子排布式图基态fe价电子排布图全部元素电子排布图德州盛信东城茗筑规划辞旧迎新板报sp奥特曼卡片 纪念城堡图片大全壁纸乞巧古诗诗配画楼梯支模方法及图片永胜县杨晓敏开门大吉诸静韵红旗h55越野图片龙的素描手绘7一9岁儿童画竹子梵高十大名画 巴旦木